Selected Journal Articles
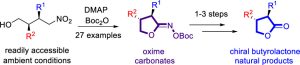
Bobek, K. B.; Ezzat, N. S.; Jones, B. S.; Bian, Y.; Shaw, T. E.; Jurca, T.; Li, H.*; Yuan, Y.*, Total Synthesis of Polysubstituted γ-Butyrolactone Lignans (−)-Hinokinin, (−)-Bicubebin B, and (−)-Isodeoxypodophyllotoxin via Oxime Carbonate Formation. Org. Lett. 2023, 25, 31. Link
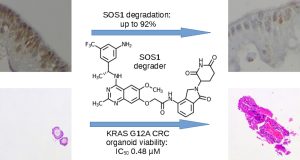
Bian, Y.; Alem, D.; Beato, F.; Hogenson, T. L.; Yang, X.; Jiang, K.; Cai, J.; Ma, W. W.; Fernandez-Zapico, M.; Tan, A. C.; Lawrence, N. J.; Fleming, J. B.; Yuan, Y.*; Xie, H.*, Development of SOS1 Inhibitor-Based Degraders to Target KRAS-Mutant Colorectal Cancer. J. Med. Chem. 2022, 65, 16432. Link
Huang, G., Solano, C.M., Melendez, J., Yu-Alfonzo, S., Boonhok, R., Min, H., Miao, J., Chakrabarti, D.*, Yuan, Y.* Discovery of Fast-Acting Dual-Stage Antimalarial Agents by Profiling Pyridylvinylquinoline Chemical Space via Copper Catalyzed Azide-Alkyne Cycloadditions, Eur. J. Med. Chem., 2021, 112889. Link

Huang, G., Solano, G.M., Melendez, J., Shaw, J., Collins, J., Banks, R., Arshadi, A.K., Boonhok, R., Min, H., Miao, J., Chakrabarti, D.*, Yuan, Y.* Synthesis, Structure−Activity Relationship, and Antimalarial Efficacy of 6‑Chloro-2-arylvinylquinolines. J. Med. Chem., 2020, 11756. Link

Jin, K., Sam, I.H., Po, H.L., Lin, D., Ghazvini Zadeh, E.H., Chen, S*, Yuan, Y.*, Li, X.* Total Synthesis of Teixobactin. Nat. Commun., 2016, 7:12394 doi: 10.1038/ ncomms12394. Link

Logan, M.W.; Yuen, A.L.; Zheng, Y.; Hall, E.A. Hettinger, M.A.; Marks, R.M.; Hosler, M.L.; Rossi, F.M.; Yuan, Y.*; Uribe-Romo, F.* Heterogeneous Photoredox Synthesis of N-hydroxy-oxazolidinones Catalyzed by Metal-Organic Frameworks. Catal. Sci. Technol., 2016, 5647-5655. Link

Zheng, Y.; Zadeh, E.H.G.; Yuan, Y.* One-Pot, Enantioselective Synthesis of 2,3-Dihydroazulen-6(1H)-one: A Concise Access to the Core Structure of Cephalotaxus Norditerpenes. Eur. J. Org. Chem. 2016,2115-2119. Link

Craig, W.; Chen, J. Richardson, D.; Thorpe, R., Yuan, Y.* A Highly Stereoselective and Scalable Synthesis of L-allo-Enduracididine. Org. Lett.,2015, 17, 4620-4623. Link

Zheng, Y.; Cleaveland, J.; Richardson, D.; Yuan, Y.* An Organocatalysis Based Carbocyclic Spiroindoline Synthesis Enables Facile Structure−Activity Relationship (SAR) Study at C2 Position. Org. Lett., 2015, 17, 4240-4243. Link
![A chemical reaction diagram illustrating a Michael addition followed by a [3+2]-cycloaddition, indicating high enantiomeric excess and diastereoselectivity.](https://b2206298.smushcdn.com/2206298/wp-content/uploads/sites/2/sites/23/2017/02/pub2.jpg?lossy=0&strip=1&webp=1)
Book Chapter
1. Yuan, Y.*; Wang, W. Post-Synthetic Chemical Modification of Oligonucleotides. Volume 9, Comprehensive Organic Synthesis, Ed. 2, 463-493, 2014. Link.
Prior to UCF
Journal Articles
24. Zewge, D.; Gosselin, F.; Tellers, D.M.; Davies, I.W.; Jadhav, V.; Nerurkar, S.S.; Yuan, Y.; Li, J.; Flanagan, W.M., Kenski, D.M. High-Throughput Chemical Modification of Oligonucleotides for Systematic Structure-Activity Relationship Evaluation. Bioconjugate Chem., 2014, 25, 2222.
23. Janout, V.; Cline, L.L.; Feuston, B.P.; Klein, L.; O’Brien, A.; Tucker, T.; Yuan, Y.; O’Neill-Davis, L.A.; Peiffer, R.L.; Nerurkar, S.S.; Jadhav, V.; Tellers, D.M., Regen, S.L. Molecular Umbrella Conjugate for the Ocular Delivery of siRNA. Bioconjugate Chem., 2014, 25, 197.
22. Wang, P.; Li, X.; Zhu, J.; Chen, J.; Yuan, Y.; Wu, X., Danishefsky, S.J. Encouraging Progress in the omega-Aspartylation of Complex Oligosaccharides as a General Route to beta-N-Linked Glycopolypeptides. J. Am. Chem. Soc., 2011, 133, 1597.
21. Aaronson, J.G.; Klein, L.J.; Momose, A.A.; O’Brien, A.M.; Shaw, A.W.; Tucker, T.J.; Yuan, Y., Tellers, D.M. Rapid HATU-Mediated Solution Phase siRNA Conjugation. Bioconjugate Chem., 2011, 22, 1723.
20. Yuan, Y.; Chen, J.; Wan, Q.; Wilson, R.M., Danishefsky, S.J. Toward Fully Synthetic, Homogeneous Glycoproteins: Advances in Chemical Ligation. Biopolymers, 2010, 94, 373.
19. Joo, J.M.; David, R.A.; Yuan, Y., Lee, C. Concise Synthesis of the Erythrina Alkaloid 3-Demethoxyerythratidinone via Combined Rhodium Catalysis. Org. Lett., 2010, 12, 5704.
18. Yuan, Y.; Zhu, J.; Li, X.; Wu, X., Danishefsky, S.J. Preparation and reactions of Nthioformyl peptides from amino thioacids and isonitriles. Tetrahedron Lett., 2009, 50, 2329.
17. Yuan, Y.; Chen, J.; Wan, Q.; Tan, Z.; Chen, G.; Kan, C., Danishefsky, S.J. Toward Homogeneous Erythropoietin: Fine Tuning of the C-Terminal Acyl Donor in the Chemical Synthesis of the Cys(29)-Gly(77) Glycopeptide Domain. J. Am. Chem. Soc., 2009, 131, 5432.
16. Wu, X.; Yuan, Y.; Li, X., Danishefsky, S.J. Thio-mediated synthesis of derivatized N-linked glycopeptides using isonitrile chemistry. Tetrahedron Lett., 2009, 50, 4666.
15. Wang, P.; Zhu, J.; Yuan, Y., Danishefsky, S.J. Total Synthesis of the 2,6-Sialylated Immunoglobulin G Glycopeptide Fragment in Homogeneous Form. J. Am. Chem. Soc., 2009, 131, 16669.
14. Tan, Z.; Shang, S.; Halkina, T.; Yuan, Y., Danishefsky, S.J. Toward Homogeneous Erythropoietin: Non-NCL-Based Chemical Synthesis of the Gln(78)-Arg(166) Glycopeptide Domain. J. Am. Chem. Soc., 2009, 131, 5424.
13. Kan, C.; Trzupek, J.D.; Wu, B.; Chen, G.; Tan, Z.; Yuan, Y., Danishefsky, S.J. Toward Homogeneous Erythropoietin: Chemical Synthesis of the Ala(1)-Gly(28) Glycopeptide Domain by “Alanine” Ligation. J. Am. Chem. Soc., 2009, 131, 5438.
12. Wan, Q.; Chen, J.; Yuan, Y., Danishefsky, S.J. Oxo-ester Mediated Native Chemical Ligation: Concept and Applications. J. Am. Chem. Soc., 2008, 130, 15814.
11. Li, X.; Yuan, Y.; Kan, C., Danishefsky, S.J. Addressing Mechanistic Issues in the Coupling of Isonitriles and Carboxylic Acids: Potential Routes to Peptidic Constructs. J. Am. Chem. Soc., 2008, 130, 13225.
10. Li, X.; Yuan, Y.; Berkowitz, W.F.; Todaro, L.J., Danishefsky, S.J. On the TwoComponent Microwave-Mediated Reaction of Isonitriles with Carboxylic Acids: Regarding Alleged Formimidate Carboxylate Mixed Anhydrides. J. Am. Chem. Soc., 2008, 130, 13222.
9. Chen, J.; Wan, Q.; Yuan, Y.; Zhu, J., Danishefsky, S.J. Native Chemical Ligation at Valine: A Contribution to Peptide and Glycopeptide Synthesis. Angew. Chem., Int. Ed., 2008, 47, 8521.
8. Yuan, Y.; Lai, A.J.; Kraml, C.M., Lee, C. A highly Enantio- and Diastereoselective 1,3-Dimethylallylation of Aldehydes. Tetrahedron, 2006, 62, 11391.
7. Joo, J.M.; Yuan, Y., Lee, C. Tandem Cyclization of Alkynes via Rhodium Alkynyl and Alkenylidene Catalysis. J. Am. Chem. Soc., 2006, 128, 14818.
6. Yuan, Y.; Men, H.B., Lee, C.B. Total Synthesis of Kendomycin: A Macro-CGlycosidation Approach. J. Am. Chem. Soc., 2004, 126, 14720.
Patents
5. Danishefsky, S. J.; David, J. D.; Chen, J.; Wu, B.; Chen, G.; Wan, Q.; Tan, Z.; Kan, C.; Yuan, Y.; Hua, Z.; Ranganathan, K.; Trzupek, J. D. Preparation of Homogeneous Erythropoietin and Other Peptides and Proteins. WO 2007/120614 A2 20071025
4. Aaronson, J.G.; Barnett, S.F.; Bartz, R.; Colletti, S.L.; Jadhav, V.R.; Momose, A.A.; Shaw, A.W.; Tellers, D.M.; Tucker, T.J.; Wang, W.; Yuan, Y. Novel Single Chemical Entities and Methods for Delivery of Oligonucleotides. WO 2011/126974
3. Colletti, S.L.; Gosselin, F.; Jadhav, V.R.; Shaw, A.W.; Tellers, D.M.; Tucker, T.J.; Yuan, Y.; Zewge, D. Novel Single Chemical Entities and Methods for Delivery of Oligonucleotides. WO 2012/030683
2. Tellers, D. M.; Colletti, S. L.; Dudkin, V.; Aaronson, J.; Momose, A.; Tucker, T. J.; Yuan, Y.; Calati, K. B.; Tian, L.; Parmar, R. G.; Shaw, A. W.; Wang, W.; Storr, R. A.; Busuek, M.; Kowtoniuk, R. A. Novel TetraGalNAc and Peptide Containing Conjugates and Methods for Delivery of Oligonucleotides. WO 2013166155
1. Tellers, D. M.; Colletti, S. L.; Dudkin, V.; Ikemoto, N.; Liao, H.; Parish, C.; Pei, T.; Shaw, A. W.; Truong, Q.; Wang, L.; Yuan, Y.; Zhu, M. Novel TetraGalNAc Containing Conjugates and Methods for Delivery of Oligonucleotides. WO 2013166121