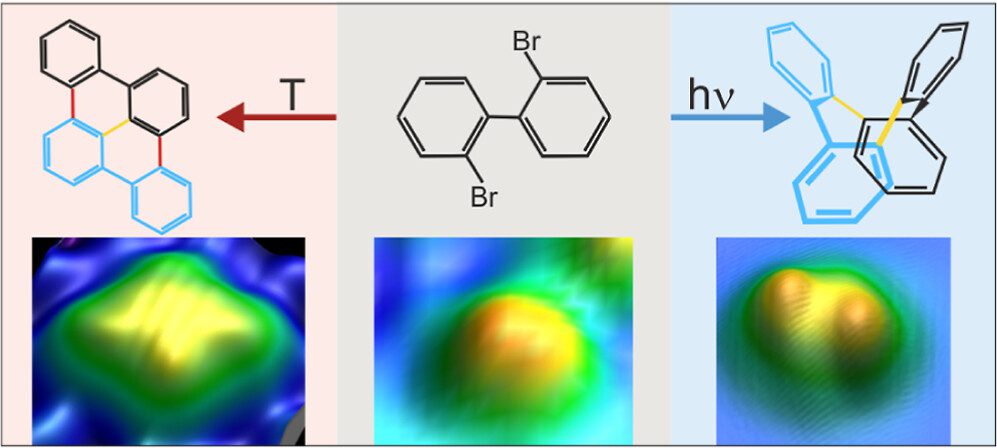
On-surface synthesis is a powerful method that has emerged recently to fabricate a large variety of atomically precise nanomaterials on surfaces based on polymerization. It is very successful for thermally activated reactions within the framework of heterogeneous catalysis. As a result, it often lacks selectivity. We propose to use selective activation of specific bonds as a crucial ingredient to synthesize desired molecules with high selectivity. In this approach, thermally nonaccessible products are expected to arise in photolytically activated on-surface reactions with high selectivity. We demonstrate for assembled 2,2′-dibromo biphenyl clusters on Cu(111) that the thermal and photolytic activations yield distinctly different products, combining submolecular resolution of individual product molecules in real-space imaging by scanning tunneling microscopy with chemical identification in X-ray photoelectron spectroscopy and supported by ab initio calculations. The photolytically activated Ullmann coupling of 2,2′-dibromo biphenyl is highly selective, with only one identified product. It starkly contrasts the thermal reaction, which yields various products because alternate pathways are activated at the reaction temperature. Our study extends on-surface synthesis to a directed formation of thermally inaccessible products by direct bond activation. It promises tailored reactions of nanomaterials within the framework of on-surface synthesis based on the photolytic activation of specific bonds.
This work was published in ACS Nano.
[C. Schunke, P. Schweer, E. Engelage, D. Austin, E. D. Switzer, T. S. Rahman, K. Morgenstern, “Increased Selectivity in Photolytic Activation of Nanoassemblies Compared to Thermal Activation in On-Surface Ullmann Coupling.” ACS Nano (2024). https://doi.org/10.1021/acsnano.3c11509]