The Soft Condensed Matter and Biological Physics group focuses on theoretical, computational, experimental, as well as clinical research. The experimental work involves various spectroscopic studies of proteins under high pressure and other extreme conditions, protein membrane interactions and light-scattering studies. The theoretical and computational studies involve both classical and quantum calculations at various length and time scales using high performance computing systems. There is a strong collaboration between the Biophysics group at the Physics department and several faculty at the College of Medicine. The research of several faculty are funded by the NSF and the NIH. The Biophysics group hosts monthly seminars typically attended by students and faculty from Physics, Chemistry, Biology, Biomedical Sciences, NanoScience Technology, and CREOL.
Read more details
Professor Alfons Schulte studies the effects of high pressure on cellular and soft matter systems. His laboratory is unique in the combination of high pressure and micro-spectroscopic techniques extending to the single cell level. He is also developing novel microspectroscopic tools such as label-free confocal absorption microscopy (two patents awarded recently) and applies them to materials characterization.

Representative publications:
- “Spatially resolved micro-absorption spectroscopy with a broadband source and confocal detection,” Arora, J. Mauser, D. Chakrabarti, A. Schulte, Opt. Comm. 355, 533-537 (2015).
- Applications of laser Raman spectroscopy,” Schulte and Y. Guo. Invited chapter in Handbook of Solid State Spectroscopy, p. 661- 688, Ed. D. R. Vij, Springer (2006).
Professor Suren Tatulian’s molecular biophysics group focuses on the structure and function of biomedically important proteins and peptides as studied by biophysical tools, such as Fourier transform infrared (FTIR) spectroscopy, fluorescence, and circular dichroism (CD). A major ongoing project aims at elucidation of the structural basis of neurotoxicity of amyloid beta peptide. In collaboration with Physics Department faculty, we study the morphology and the atomic-resolution structure of protein assemblies using transmission electron microscopy (TEM), atomic force microscopy (AFM), and nuclear magnetic resonance (NMR).
The cytotoxic effect of amyloid beta peptide of defined structure is studied in collaboration with our colleagues from UCF College of Medicine. These studies have a long-term goal of identification of new biomarkers for development of advanced diagnostic and therapeutic strategies to combat Alzheimer’s disease.

Representative publications:
- Tatulian S. A. (2016) Interfacial enzymes: Membrane binding, orientation, membrane insertion, and activity. In: Enzymology at the Membrane Interface (Gelb, M. H., ed.), Ch. 10, Elsevier (in press).
- Goldblatt , Matos J.O., Gornto J., and Tatulian, S.A. (2015) Isotope-edited FTIR reveals distinct aggregation and structural behaviors of unmodified and pyroglutamylated amyloid β peptides. Phys. Chem. Chem. Phys. 17, 32149–32160
- Matos, J.O., Goldblatt, G., Jeon, J., Chen, B., and Tatulian, S.A. (2014) Pyroglutamylated Amyloid-b Peptide Reverses Cross b-sheets by a Prion-like Mechanism. Phys. Chem. B 118(21), 5637-5643
Professor Bo Chen’s research group combines experimental and theoretical biophysics methods to investigate the structure, dynamics and function correlation of self-assembled proteins. Specifically, the structure of proteins in their functional assembled states is characterized by nuclear magnetic resonance (NMR) and transmission electron microscopy (TEM). Based on the experimental structural information, suitable models are constructed to understand the assembly mechanism and to reveal the structural function correlation. Current projects include the HIV and Rous sarcoma viral capsid protein and amyloid-beta proteins implicated in Alzheimer’s diseases.
Representative publications:
- Jaekyun Jeon*, Ivan Hung, Alok K. Mitra, Ambroise Desfosses, Xin Qiao*, Daniel Huang*, Peter L. Gor’kov, Rebecca C. Craven, Richard L. Kingston, Zhehong Gan, Fangqiang Zhu, and Bo Chen. “Structural model of the tubular assembly of the rous sarcoma virus capsid protein”, JACS, in press.
The link to the paper is http://pubs.acs.org/doi/pdf/10.1021/jacs.6b11939. DOI: 10.1021/jacs.6b11939 - Xin Zhao*, Jaekyun Jeon*, Jeff Weber*, and Bo Chen. “Mechanism of polymorphism and curvature of HIV capsid assemblies probed by 3D simulations with a novel high resolution coarse grain model”, BBA-Gen Subjects, 1850(11):2353-67(2015).
- Jaekyun Jeon*, Michael S. Lodge, Ben D. Dawson, Masa Ishigami, Frank Shewmaker, and Bo Chen, BBA – General Subjects, 1830, 3807-3815 (2013). “Superb resolution and contrast of transmission electron microscopy images of non-stained biological samples on graphene-coated grids”.
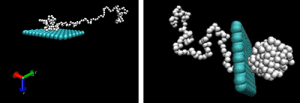
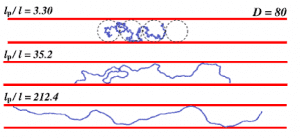
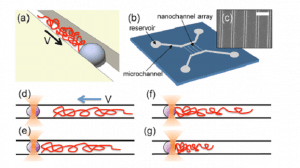
Dr. Aniket Bhattacharya uses scaling theory of polymer physics and a variety of numerical methods to study conformations and dynamics of DNA and other biopolymers under confinement. Most recent activities include investigations of DNA transportation through protein pores, confinement of DNA inside nano-channels.
Representative publications:
- Kaifu Luo, Tapio Ala-Nissila, S-C Ying, and Aniket Bhattacharya, “Sequence dependence of DNA translocation thorough a nanopore”, Phys. Rev. Lett. 100 1058101 (2008).
- Aiqun Huang, H.-P Hsu, Aniket Bhattacharya, and Kurt Binder, “Semiflexible macromolecules in quasi-one-dimensional confinement: Discrete versus continuous bond angles”, J. Chem. Phys. 143, 243102 (2015). (Invited article)
- Aiqun Huan, Walter Reisner, and Aniket Bhattacharya, “Dynamics of DNA Squeezed inside a Nanochannel via a Sliding Gasket” (invited) Polymers 2016, 8(10), 352; doi:10.3390/polym8100352.

